www.elsevier.es/rmuanl
ORIGINAL
ARTICLE
Evaluation
of
the
criteria
for
the
interpretation
of
the
oral
glucose
tolerance
test
in
the
National
Mother-Child
Teaching
Hospital
‘‘San
Bartolome’’
J.J.
Moya-Salazar
a,∗,
L.
Pio-Dávila
baHospitalNacionalDocenteMadre-Ni˜no‘‘SanBartolomé’’,Lima,Peru bHospitalNacionalArzobispoLoayza,Lima,Peru
Received9March2015;accepted28April2015 Availableonline18August2015
KEYWORDS Diabetesmellitus; Glucosetolerance test;
Glucose
Abstract Diabetesmellitus(DM)isametabolicdisorderthatcauseschronichyperglycemia with disturbancesinthemetabolism ofcarbohydrates,fatsandproteins,andalterationsin microcirculation.Weevaluatethecriteriafortheinterpretationoftheoralglucosetolerance test(OGTT)intheHospitalNacionalDocenteMadre-Ni˜no‘‘SanBartolomé’’inLima,Peru,and determinethepercentagesofpre-diabeticpatients,diabeticsthatshouldnotbeincludedinthe test,andthosewithalterationsintheirglucosecurveduringthebiochemicaldeterminations. To this purpose, a non-experimental, prospective cross-sectional analytic study was performedin1271patients,includedinthestudytocomplywiththeguidelinesand recommen-dationsoftheADAandtheCLSIPOCT12-A3guide,whichwereprocessedintheBiochemical AutoanalyzerBiosystemsA25.DataanalysiswasperformedusingSPSSversion20.0statistical analyzer,andrespectingethicaldiabetics.
Themainresultsare1.97%diabeticand13.85%pre-diabetic.TheLJphenomenonoccurred with6.45%(alterationinglucosecurveafteroralloaddecreasesplasmaglucoseconcentration determinationafter60minandreturnsatrelativelyhigherthanthebasalconcentrationlevels at 180min)and5.19% were poorlystudied, includinga>110mg/dl baseline(p=<0.05).We determinedahighrateofpre-diabeticpatientsandareducedrateofdiabetescoincidentwith Qiaoetal.WehighlighttheusefulnessoftheOGTT,whichmustberigorouslyevaluated,and reaffirm theimportanceoftheLJ phenomenonasitmay interferewiththe finalresultsof unsolvedevents,andevidencetheimportanceofinterindividualbiologicalvariability. © 2015 Universidad Autónoma de Nuevo León. Published by Masson Doyma México S.A. ThisisanopenaccessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/ licenses/by-nc-nd/4.0/).
∗Correspondingauthorat:Departamentodeayudaaldiagnóstico,HospitalNacionalDocenteMadre-Ni˜no‘‘SanBartolomé’’,Jr.Pacifico
957,Lima07,Lima,Peru.Tel.:+511986014954.
E-mailaddress:jeelms@outlook.com(J.J.Moya-Salazar).
http://dx.doi.org/10.1016/j.rmu.2015.04.004
Introduction
Diabetes mellitus (DM) is a metabolic disorder that is a consequence ofthe deficiency in thesecretionof insulin, in the effectiveness of its actions, or both. It is one of themost prevalentchronic diseases in theworld. It con-stitutes a public health problem due to the progressive increaseof its incidence,tothe point where itis consid-eredanepidemic.1Chronichyperglycemiawithdisturbances
inthemetabolismofcarbohydrates,fatsandproteins,and alterationsinmicrocirculationareallconsequencesofthis disorder.CurrentDMclassificationaccordingtothe Ameri-canDiabetesAssociation---ADA---isshowninTable1.
About63%ofthe57milliondeathsintheworldin2008 werecausedbyanon-transmissiblechronicdisease, includ-ingdiabetes mellitus; thus, 80% of thesedeaths occurred in low and medium-income countries.2 By the year 2000,
about171millionpeoplesufferedfromDMaroundtheworld, andthisnumberisestimatedtogoupto336millionbythe year2030.3,4Ontheotherhand,approximately197million
people worldwide suffer from glucose intolerance --- pre-diabeticpatients,whichusuallyleadstoobesity.Around90% oftypeIIdiabetesisattributabletooverweightnessandto themetabolic syndrome.This numberis alsoexpected to increaseto420millionbytheyear2025.5Inaddition,the
humanandfinancialcostsofDMarealsoontherise.6
A similar situationoccurs in Peru, where TIIDM preva-lence(TIIDM)rangesfrom1to8%,LimaandPiurabeingthe mostaffectedregions.7Inconclusion,DMisahealthproblem
aroundtheworld,onethatisthreateningtoreachpandemic levelsby2030,withalarmingincreasesofTIIDMamong chil-drenandwithpotentiallydevastatingconsequences.5
DMis associated withan increase in risk of premature death;thus,everyyearalmost4milliondeathsarecaused directlybythisdisease.80%oftheseoccurin underdevel-opedcountries,constituting6.8%ofoverallmortality.8
DM diagnostic methods are: random glucose test >200mg/dlover clinicalsymptoms,fastingplasma glucose test>126mg/dl,oralglucosetolerancetest(OGTT)aftera fast,andglycosylatedhemoglobintestA1c≥6.5%(Table2).9
ThetestmostfrequentlyusedistheOGTT,usedinclinical practicefor glycemicandinsulin diagnoses.10---12 Moreover,
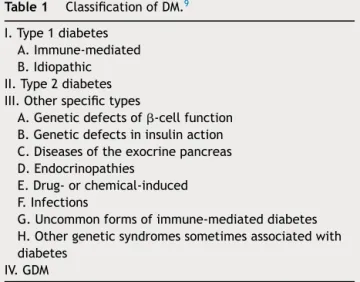
Table1 ClassificationofDM.9
I.Type1diabetes A.Immune-mediated B.Idiopathic II.Type2diabetes III.Otherspecifictypes
A.Geneticdefectsof-cellfunction B.Geneticdefectsininsulinaction C.Diseasesoftheexocrinepancreas D.Endocrinopathies
E.Drug-orchemical-induced F.Infections
G.Uncommonformsofimmune-mediateddiabetes H.Othergeneticsyndromessometimesassociatedwith diabetes
IV.GDM
Table2 DiagnosticcriteriaforDM.9
1.AlC≥6.5%.Thetestshouldbeperformedinalaboratory usingamethodthatisNGSPcertifiedandstandardizedto theDCCTassay.a
OR
2.FPG≥126mg/dl(7.0mmol/l).Fastingisdefinedasno caloricintakeforatleast8h.a
OR
3.2-hplasmaglucose≥200mg/dl(11.1mmol/l)duringan OGTT.Thetestshouldbeperformedasdescribedbythe WorldHealthOrganization,usingaglucoseload containingtheequivalentof75ganhydrousglucose dissolvedinwater.a
OR
4.Inapatientwithclassicsymptomsofhyperglycemiaor hyperglycemiccrisis,arandomplasma
glucose≥200mg/dl(11.1mmol/l).
a Intheansenceofunequivocalhyperglycemia,criteria1---3
shouldbeconfirmedbyrepeattesting.
it is the most sensitive method for the diagnosis of DM withinthefastingglycemictest,althoughitrequires char-acteristics for its implementation, such as fasting basal glycemia<110mg/dl, doses of 75g anhydrous glucose in adultsor1.75g/kgforchildren,performanceinthe morn-ingaftera10---16hfast,thedurationofthetestis180min, etc.13 Oneofthemain disadvantagesofOGTTisits
repro-ducibility,thusitisrecommendedtohaveatleast2 patho-logicalOGTTswhenbasalglycemiaisunder110mg/dl.14
Moreover, inter-individual variabilities have been describedaccordingtoglucosemetabolism,whichgenerate slantsintheinterpretationofclinicalcriteriathatoughtto be estimated. According to Clinical Laboratory Standards
Institute (CLSI),the resultant parametersof OGTTshould
beassessedastheareaunderthecurveofglucose(AUCG) and insulin (AUCI) basal indexes, the maximum allowable error,amongothers,mainlytodeterminethesensitivityto peripheral insulin.9,15---18 After an oral glycemia overload,
theincreaseinglycemiadoesnotdependsolelyonglucose. There are also intestinal hormones involved, the speed of gastric emptying and the composition of the intake. Thus, tolerance to glucose and sensitivity to insulin are different concepts, making the indexes of correction and evaluation of glycemic sensitivity necessary to guarantee the quality of the results and explain the recurrence of thesephenomena.18
Havingsaidthat,theobjectiveofthisstudywasto eval-uatethecriteriaforinterpretationofOGTTintheHospital NacionalDocenteMadre-Ni˜no‘‘SanBartolomé’’ (HONADO-MANI SB) in Lima, Peru, and determine the percentages of pre-diabetic patients,thosewho shouldnothave been included within the test (basal glucose>110mg/dl) and those who presented alterations in the curve of glucose duringbiochemicaldeterminations.
Materials
and
method
Populationandsample
Population
Thepopulationconsistedofallambulatorypatientsreferred tousbyagreement,atHONADOMANISB.
Sample
Blood samples referred from outpatient clinics indepen-dently from the Department of Help to diagnosis at the Biochemistry Department for glycemia determination though OGTT, which complies with the quality criteria according to CLSI Guide POCT12-A3, the ADA’s guidelines and the recommendations for the laboratory analysis of DMdiagnosisandmanagementandthestandardized oper-ationalprocedures---SOP---ofthehealthcenter.19,20These
areselectedrespectingthefollowingpreviouslyestablished criteriaofinclusionandexclusion.
Inclusioncriteria. Patients(maleandfemale)inafasting state of at least 10h, ages ranging from 18 to 65 years ofage, stable,without astress condition,pharmaceutical consumption, or debilitating situations like surgery. Also, patients were required to complete the test (180min, 3 bloodcollections)post-glycemicoverload.Thecollectionof samplesmustcomplywiththequalityguidelinespreviously mentioned.
Exclusioncriteria. Patientswhodidnotcomply withthe
fasting state of at least 10h. Patients outside the age range(19---65 yearsold).Patientswhoweresufferingfrom adebilitatingoroncologicalsituation,orundergoing phar-macologicaltherapies. Thosepatientswholeftthetestor complicated blood collections.Those sampleswhich were collected without following the quality and normativity criteriaofCLSIor ADA,inadditiontosampleswhichwere clearlycontaminated.
Datacollectiontechniquesandsampleprocessing
Pre-analyticstage
The collection wasconducted in the Phlebotomyarea of the HONADOMANI SB between 6am and 9am, using BD Vacutainer®(FranklinLakes,NewJersey,USA)of3ml,with
a red cap which was mixed by inversion of 8---10 times, accordingtotheCLSIH03-A6guide.Inordertoachievethis, patientsmusthaveremainedfastingforatleast10hprior
tothecollection,withapaymentslipfromthepreviousday ofthetest,andwithalemonandadisposablecup.21
Thefirstsample(basal)wasusedtoevaluatethe admis-sion of patients to the test, excluding those who were above100mg/dl.Subsequently,theywereoverloadedwith oralglucose(anhydridepreparation:‘‘lemonade’’)usingthe followingformula; 75g of anhydrideglucosefor adults or 1.75g/kg forchildren. The patientwasmonitoredfor the whole duration of the test (180min). The samples were collected at 60 and 180min after the glycemic overload. Conventional cohort values were used at >200mg/dl for diabeticsandthoseindicatedbytheWorldHealth Organi-zation(WHO) forthe diagnosis ofpre-diabeticpatients of 100---199mg/dl(impairedglucosetolerance).22---24
Analyticstage
The processing in the clinical lab was conducted follow-ing the algorithm established by the hospital for OGTT. This includes the reception and registration of the sam-plethroughthestandardizedcodeanditsprocessingwithin 120min after the sample was drawn. The biochemical processingwasconductedthroughtheBiochemical Biosys-tems A25 auto-analyzer (Pennsylvania, USA), which has a daily and historic registry of biochemical analyses. The methodemployedforthedeterminationofglucosewas glu-coseoxidase-peroxidase throughthe glucose activity test Trinder.25
Post-analyticstage
Undeterminedresultsorthosewhichwereoutsidethe lin-earityofthetrialwererepeatedand/ordiluted.Theresults validatedbyMedical Technologistswereinputted intothe integratedhealthsystem---SIGOS---toinformpatientswithin thestipulatedtimes.
Dataanalysistechnique
Data analysis was performed in three basic processes: codification, from the Biochemical Biosystems A25 auto-analyzer’shistoricalregistriessystem;tabulation,statistical verificationandthecreationofchartsandtablesusingthe statisticalanalyzerSPSS20.0andMicrosoftOfficeExcel2010 forWindows.Theevaluationofthedistributionofvariables wasconductedusingKMO andBartlett’stest ofsphericity, resultinginamatrixofcorrelationsbetweenadequate varia-bles(p=<0.05).
Assessments OGTT
0.00% 2.00% 4.00% 6.00% 8.00% 10.00% 12.00% 14.00%
Prediabetics
LJ phenomenon
Basal glucose >110 mg/dl
Diabetics
Ethics
Within the diabetic ethics framework, the safeguarding, reliabilityandirreplaceablevalueoftheobtained informa-tionwillonly beusedfor the purposesof this study.This researchhastheapprovaloftheDepartmentofTeachingand AidforResearchandtheEthicsandResearchCommissionof theHONADOMANISB(Reg.78-2015).
Limitations
Several limitations should be taking into account before interpretingtheresults.
First, the biochemical auto-analyzer informatics reg-istry does not have detailed registries or information of the selected patients, such as age, gender, attributable risk factors, physiological condition, diseases, demo-graphic characteristics and family history, etc. Second, thedeterminationofglycemiavariesconsiderablybetween demographicsand age groups. Thus,the pre-diabeticand diabeticprevalence ratesencountered arenottransposed toward the general population of Peru. Third, the OGTT results could not be compared to the standard refer-ence tests like Hb1Ac, to determine its variability and inaccuracy.26 Lastly, the biochemical processing was
per-formed under an internal and external quality control; however,withoutaqualityresponsibleplanning.
Despitetheselimitations,ourresearchisthefirstto eval-uatetheresultantparametersoftheOGTT.
Results
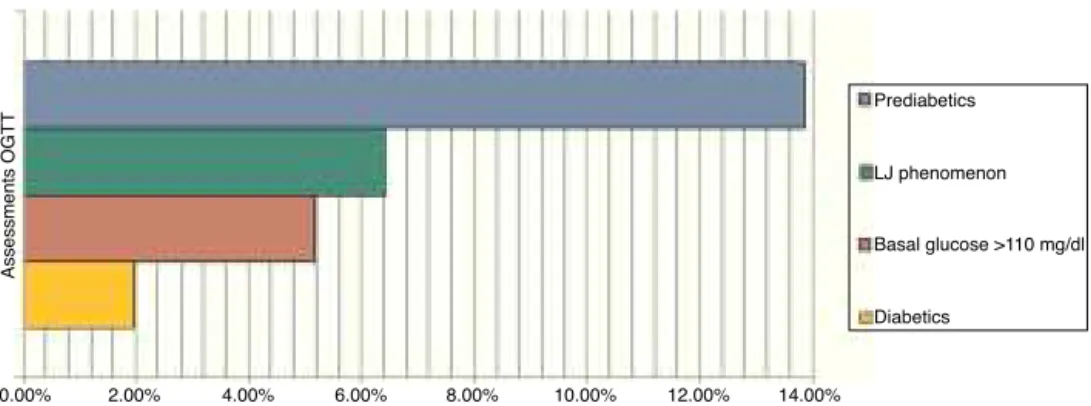
From the conducted research, 1.97% of patients were diabetic, 13.85% were pre-diabetic, 6.45% produced the ‘‘LowJump’’(LJ)glucosereduction-risephenomenon (alter-ationofthe glucosecurve post-oraloverload whichtends toreducetheconcentrationofplasmaglucoseinits deter-minationat60minandreturnstorelativelysuperiorlevels tothe basal concentration at 180min), and 5.19% of the patients should not have been included in the study due tothefactthattheydidnothaveover110mg/dlofbasal glucose(p=<0.05)(Fig.1).11,27
Discussion
Theevaluationof OGTTinterpretationcriteriaexposesan elevatedrate ofpre-diabetic patientsand areducedrate ofdiabetics, whichin conjunctionconstitute15.8% ofthe prevalence(Fig.2).
The prevalence ofdiabetic patientsdetermined inthis researchwashalfofthatcommunicatedinthelastreport of DM prevalence in Lima and Callao, Peru (3.9%).28 The
prevalence rate discovered is somewhat similar to urban averages in populations over 3000asl. (Huaraz --- 1.3%).23
DMisachronic,degenerative,progressivebutmanageable disease.Ithasagreatimpactontheeconomyofthe health-care system. It requires serious control and a reasonable stratificationofitssystemiccomplications.
In the sameway, the proportionof patients whowere pre-diabetic or had a high risk of diabetes or glucose intolerance was 13.85%; prevalence within the regional average is between 5 and 15%.29 Glucose intolerance is
a risk factor for the development of TIIDM and implies a
13.85%
1.97%
Diabetic
Prediabetics
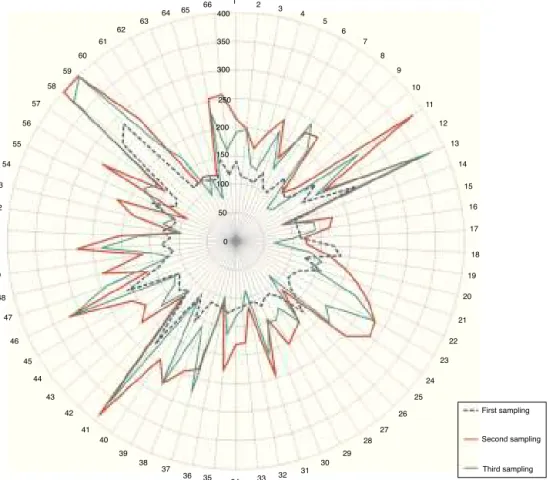
Figure2 Percentagesofpre-diabeticanddiabeticpatients. Theradialdistributionofpatientswronglyincludedinthetest isshowninFig.3,highlightingthevaluesofthesecondglycemia samplingincomparisontothefirstandthethird.
high cardiovascular risk.30,31 In our country, the reported
prevalence is up to 90.8% in patients who are 50 years and older, a highly elevated prevalence, suggesting the immediateprioritization ofhealth care attention inorder toavoidfuturecomplications.32
The progression from normoglycemia to diabetes may take several years, which involves intermediatestages of dysglycemia. This atero-thrombogenic alteration becomes evident with the alteration of glucose when fasting, emergence of glycated proteins and progressively cel-lular hypofunction, increasing the risk of morbidity in patients.33,34 Thus, diabetes indicates a decrease in the
pancreatic reserve (up to 50% when there is no diabetic manifestation),thispreviousmetabolicstageisevidentby glycemia between 100 and199mg/dl by the OGTT, which servesasaredflagtoavoiditsprogressiontoDM.24
Inter-ventioninpre-diabeticpatientsisanefficientstrategy,since it avoids or slows down the progressive deterioration of thepancreas.Thisstrategyincludesanefficientdiagnosis, modificationinlifestyles,bodyweightmanagement,ranges from45to65%ofdailyenergeticintake,physicalexercise, pharmacologicaltreatment,detectionalgorithms,etc.18 In
thismanner,itisdemonstratedandconfirmedthatthe con-trolofglycemiaisaneffectivemeasuretoreducetheload ofmicrovascularandcardiovascularcomplications,suchas retinopathy, nephropathyandneuropathy in patientswith TIDMaswellasTIIDM.35---37
1 2 3
4 5
6 7
8
9
10
11
12
13
14
15
16
17
18
19
20
21
22
23
24
First sampling
Second sampling
Third sampling 25
26
27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50
51 52 53 54
55 56
57 58
59 60
61 62
63
64 65 66400
350
300
250
200
150
100
50
0
Figure3 RadialdistributionofpatientswronglyincludedintheOGTT.Observetheelevatedvaluesinthesecondsampling(red line/thickerline).Glycemicconcentrationisexpressedinmg/dl.(Forinterpretationofthereferencestocolorinthisfigurelegend, thereaderisreferredtothewebversionofthisarticle.)
are standardized, and follow internationally established flowchartswhichruleunderaqualitycontrolsystem. More-over, the diagnosis of over half of these patients was dysglycemia(3.2%),1.5%werediabeticsandonly0.5%were healthypatients.9,11,12,15,16Consideringthat,thesemistakes
inpatientselectionarearesultofthegenerallypoor knowl-edgeof thepersonnelonthese investigations,work over-load,orthefactthatthereisnootherdiagnosticmethod. Thus, the modification of inclusion criteria of patients in theOGTT,POE,shouldbeconsideredandrelyontheCLSI POCT12-A,ClinicalLaboratoryImprovementAmendments, (CLIA),ADAguidelines,etc.Also,theuseofother diagnos-tic methods (i.e. the A1c Glycosylated Hemoglobin test), evaluatingtheirsensitivity,disadvantagesandcosts.
Lastly,wereaffirmtheimportanceoftheLJphenomenon intheinterpretationoftheOGTT,becauseitcouldinterfere in the final results. From the final reports of this phe-nomenon1.2%weredysglycemicinrangesof100---175mg/dl (Fig.1),whichleads ustoconsideritsinvolvementin the developmentof the trial and interferenceswithglycemia --- increasing it or reducing it --- generating undetermined results,orevenworse,falseresults.24
This finding proves the importance of inter-individual biologicalvariability,evident bytheevents throughwhich it may occur, like different physiological conditions, metabolismofthesaturationofglycemia,typeofanhydride
glucose,population group, diabetics, physiological condi-tionswheretypologiesoftheindividuals,pre-diabetics,or other non-clear situations occur. Thus, it is necessary to evaluate its reach in interference, to establish its total allowableerror andunderstand the biochemistryand cel-lularbehavior during the developmentof the test among individuals.
Furthermore,ajudiciousmonitoringisrequiredinorder to understand and know its physiology and the events through which it occurs, corresponding to the different metabolisms during thetest and theirdiagnostic implica-tions.Wedefinitelysuggestthatotherglycemiatestsshould beconductedinapatientwherethisphenomenonis discov-ered,(Hb1A1c,etc.)orrepeattheOGTTtwice.11
Diabetesmellitusisametabolicdiseasewhichimposesa higheconomicandsocialcostaroundtheworld,henceits preventionandtreatmentshouldbeconsideredimperative inhealthandapriorityworldwide.Thediagnosisshouldbe thoroughlyexamined,fromtheselectionofpatientstothe determinedrangesof glucoseineach collection,withthe purposeofavoidingundeterminedresultsorfalsenegatives.
Funding
Conflict
of
interest
Theauthorsstatethattherearenoconflictsofinterests.
References
1.RodbardHW,BlondeL,BraithwaiteSS,etal.American Associ-ationofClinicalEndocrinologistsmedicalguidelinesforclinical practice for the management of diabetes mellitus. Endocr Pract.2007;13Suppl.1:1---68.
2.World Health Organization. Global status report on non-communicablediseases2010.Geneva:WHO;2011.
3.International Diabetes Federation, IDF. Diabetes atlas 2012 update.5thed.Brussels:IDF;2013.
4.WildS,RoglicG,GreenA,SicreeR,KingH.Globalprevalenceof diabetes:estimatesfortheyear2000andprojectionsfor2030. DiabetesCare.2004;27:1047---53.
5.Hossain P, Kawar B, El Nahas M. Obesity and diabetes in the developing world: a growing challenge. N Engl J Med. 2007;356:213---5.
6.The world health report 2006: working together for health. Geneva:WorldHealthOrganization;2006.
7.GarcíaF,SolísJ,CalderónJ,etal.Prevalenciadediabetes mel-litusyfactoresderiesgorelacionadosenunapoblaciónurbana. RevSocPerMedInterna.2007;20:90---4.
8.Rawal RB, Tapp RJ, Williams ED, Chan C, Yasin S, Olden-burg B. Prevention oftype 2 diabetesand itscomplications indevelopingcountries:a review.IntJBehavMed.2012;19: 121---33.
9.AmericanDiabetesAssociation.Diagnosisandclassificationof diabetesmellitus.DiabetesCare.2014;37Suppl.1:S81---90.
10.MatsudaM,DeFronzoRA.Insulinsensitivityindicesobtained fromoralglucosetolerancetesting:comparisonwiththe eug-lycemicinsulinclamp.DiabetesCare.1999;22:1462---70.
11.StumvollM,MitrakouA,PimentaW,etal.Useoftheoralglucose tolerancetesttoassessinsulinreleaseandinsulinsensitivity. DiabetesCare.2000;23:295---301.
12.CalvarCE,BengoleaSV,HermesR,LoyatoM.Criticalevaluation oftheoralglucosetolerancetestforthediagnosisofinsulin resistanceinpatientswithpolycysticovarysyndrome.Medicine (Baltimore).2007;67:2---6.
13.Puig-DomingoM,LeivaHidalgoA.Diabetesmellitus:concepto, clasificaciónyetiología.In:CasanuevaFreijoF,VázquezGarcía JA,editors.Endocrinologíaclínica.DíazDeSantosEds,vol.14. 1995.p.241---9.
14.McDonaldGW,FisherGF,BurnhamC.Reproducibilityoftheoral glucosetolerancetest.Diabetes.1965;14:473---80.
15.Myllynen P, Koivisto V, Nikkila E. Glucose in tolerance and insulinresistanceaccompanyimmobilization.ActaMedScand. 1987;222:75---81.
16.MatthewsDR,Hosker JP, RudenskiAS,Naylor BA,TeacherDF, Turner RC. Homeostasismodelassessment: insulinresistance and b-cell function from fasting plasma glucose and insulin concentrationinman.Diabetología.1985;28:412---9.
17.AvignonA,BúgnerC,Mariano-GoulartD,ColetteC,MonnierL. Assessmentofinsulinsensitivityfromplasmainsulinand glu-coseinthefastingorpostoralglucose-loadstate.IntJObes. 1999;23:512---7.
18.Girbés BJ. Methods for the determination of insulin sensi-tivity based on an oral glucosetolerance test. Av Diabetol. 2008;24:296---304.
19.ClinicalLaboratoryStandardsInstitute.Point-of-careblood glu-cosetestinginacuteandchroniccarefacilities.ApprovGuidel. 2013;33:14---26.
20.American Diabetes Association Guidelines. Recommendations forlaboratoryanalysisinthediagnosisandmanagementof dia-betesmellitus.DiabetesCare.2011;34:e62---84.
21.ClinicalandLaboratoryStandardsInstitute.H3-A6.Procedures forthecollectionofdiagnosisbloodspecimensbyvenipuncture; 2007.p.27.
22.AmericanDiabetesAssociation.Screeningfortype2diabetes. DiabetesCare.2004;27Suppl.1:S11---4.
23.PanAmericanHealthOrganization.GuíasALADdediagnóstico controly tratamientode ladiabetesmellitus tipo2,vol. 3; 2006.p.12---7.
24.AmericanDiabetes Association. Standardsof medicalcare in diabetes---2014.DiabetesCare.2014;37Suppl.1:S14---20.
25.Trinder P. Determination of blood glucose using an oxidase---peroxidase system with a non-carcinogenic chromogenic.JClinPathol.1969;22:158---61.
26.American Diabetes Association. Translating the A1C assay into estimated average glucose values. Diabetes Care. 2008;31:1473---8.
27.Amatuzio DS, Stutzman LF, Vanderbilt JM, Nesbitt S. Inter-pretation ofthe rapidintravenous glucosetolerance test in normalindividualsandinmilddiabetesmellitus.JClinInvestig. 1953;32:428---35.
28.RevillaL,LópezT,SánchezS,YasudaM,SanjinésG.Prevalence ofhypertensionanddiabetesinresidentsfromlimaandCallao, Peru.RevPeruMedExpSaludPublica.2014;31:437---44.
29.Qiao Q, Lindström J, Valle TT, Tuomilehto J. Progression to clinicallydiagnosed andtreated diabetesfrom impaired glu-cosetolerance and impaired fasting glycaemia. Diabet Med. 2003;20:1027---33.
30.Levitzky YS, Pencina MJ, D’Agostino RB, et al. Impact of impaired fasting glucose oncardiovascular disease: the Fra-minghamHeartStudy.JAmCollCardiol.2008;51:264---70.
31.Vargas-AnconaL.Epidemiologiadeladiabetesmellitus, intoler-anciaalaglucosayfactoresderiesgoaterogénicoenYucatán, México.RevBiomed.1994;5:151---9.
32.Figueroa ML. Diagnóstico de Intolerancia a la glucosa en pacientes mayoresde50A˜nos, en elservicio delaboratorio delHospitalSuárezAngamosII---EsSaludLima-Perú.RevMéd. 2011;1:72---7.
33.FloresJA,DíazA,Trevi˜noS,BrambilaEM.Theeffectsofa hyper-caloricdietonthedevelopmentofdysglycemiaanddyslipemia itsimpactonthekidneycytoarchitecture.MensajeBioquímico. 2014;61:165---74.
34.ClementS, BraithwaiteSS, MageeMF,et al. Managementof diabetesand hyperglycemia in hospitals,American Diabetes Association.DiabetesCare.2004;27:553---91.
35.TheDCCTResearchGroup.Diabetescontrolandcomplications trial (DCCT): results of feasibility study. Diabetes Care. 1987;10:1---19.
36.Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes. Lancet. 1998;352(9131):837---53.