TítuloSynthesis of bench stable solid triorganoindium reagents and reactivity in palladium catalyzed cross coupling reactions
Texto completo
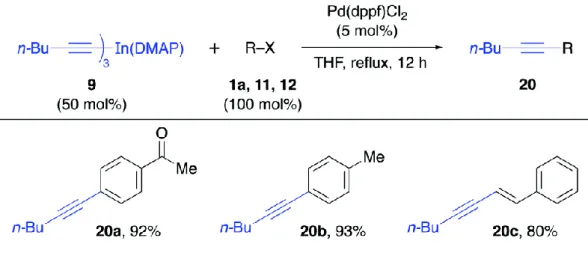
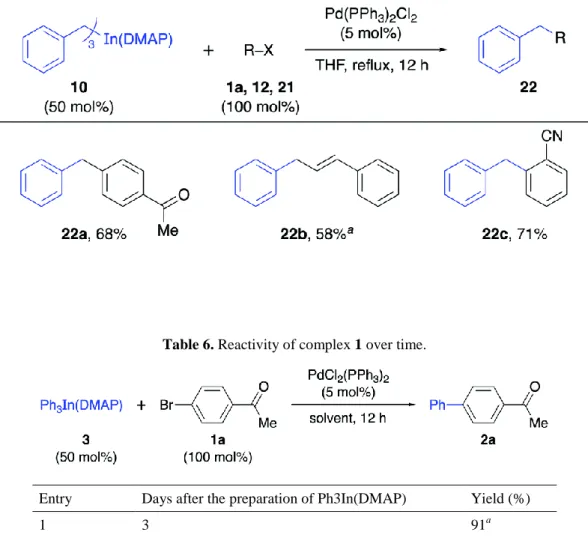
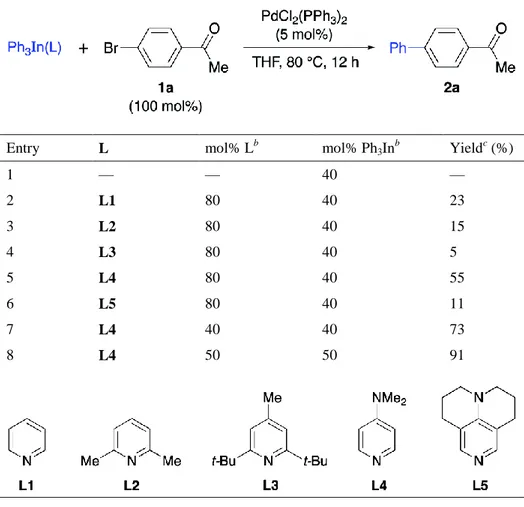
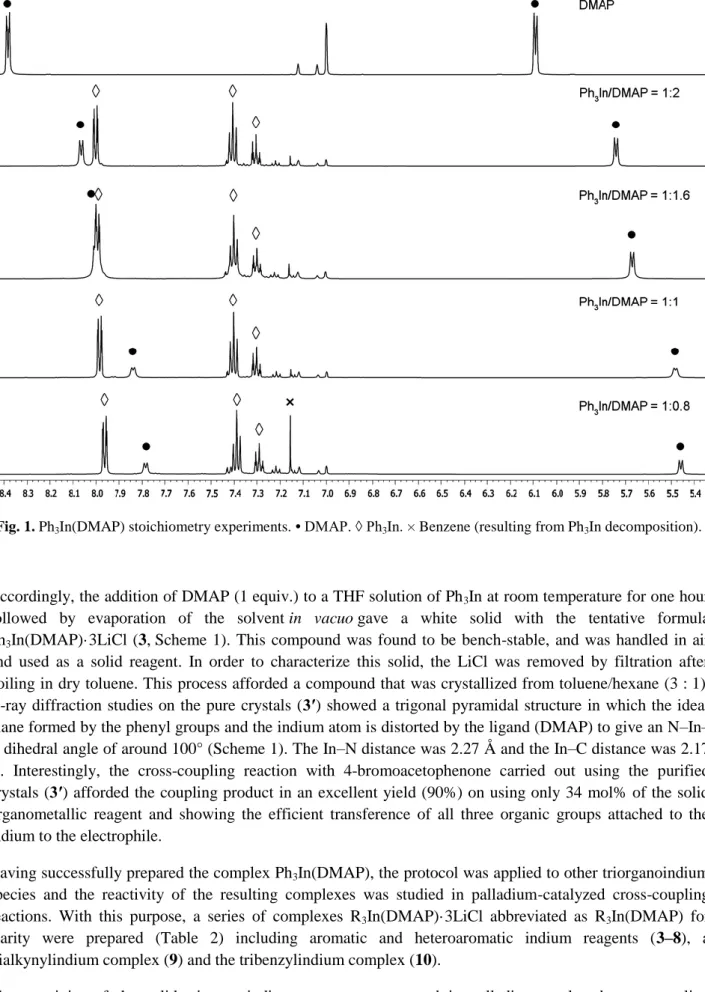
Figure
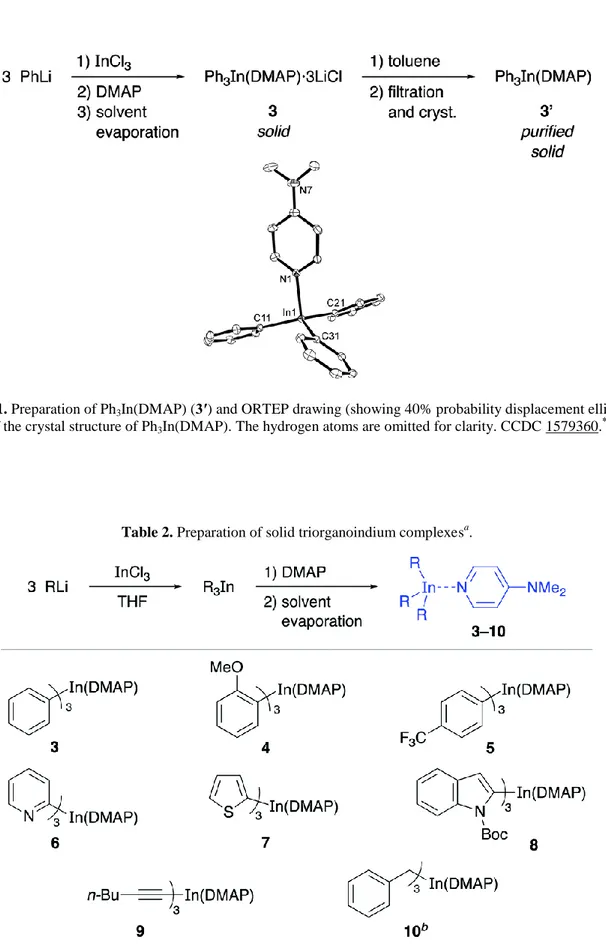
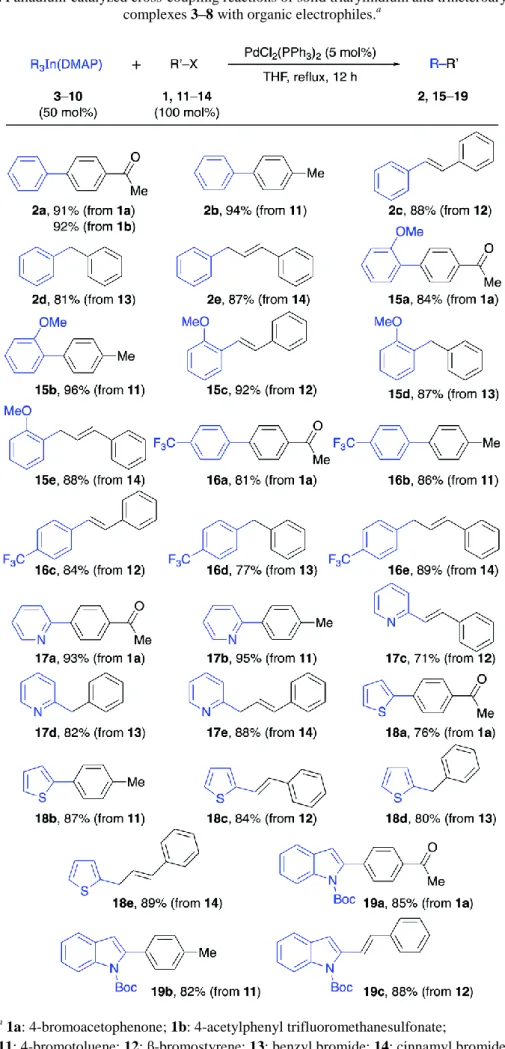
Documento similar
Jia and coworkers have described a concise total synthesis of dictyoden- drins B and C, utilizing palladium-catalyzed Larock annulation for the con- struction of the highly
The intro- duction of the element yttrium (ZYF2M2) provides a solid solution of zirconia stabilized in tetragonal form, even after calcinations at 1473 K.. Materials obtained
Herein we describe a robust and practical method to prepare enantiomerically enriched trisubstituted allenes using alkyl Grignard reagents and bench stable
active esters, which have been successfully used in simple Ni- catalyzed cross-coupling reactions, [7] and to take advantage of the presence of an alkene to realize a cyclization,
Fidelity of Human Immunodeficiency Viruses Type 1 and Type 2 Reverse Transcriptases in DNA Synthesis Reactions using DNA and RNA Templates... Universidad Autónoma de Madrid
Moreover, the enallene 70 used by Bäckvall and coworkers, 241f was also subjected to the reaction conditions giving rise to a mixture of three alkylboronates (71) located around
Ammonium salts with aryl and silyl substitution on the alkyne and different alkyl chains at the propargylic position afford- ed the desired compounds in high yield and
Today, strong coupling CQED is indeed routinely applied to a plethora of solid-state systems such as quantum dots in dielectric and metallic nanocavities and, most recently,