Scientific note/Nota científica
IR – spectroscopic characterization of biominerals
in marattiaceaeus ferns
EnRiquE J. BaRan
1,3and CRistina H. RollERi
2(received: april 17, 2008; accepted: July 01, 2010)
ABSTRACT – (iR – spectroscopic characterization of biominerals in marattiaceaeus ferns). Frond samples of the eusporangiate ferns Marattiaceae genera Angiopteris, Christensenia, Danaea and Marattia were investigated by infrared spectroscopy, under
different experimental conditions. The results conirmed the previously reported accumulation of biogenic silica (SiO2) in
tissues of these ferns and also showed, for the irst time, the presence of calcium oxalate in this group of plants, probably as
weddellite. the ability to biomineralize sio2, to produce and accumulate biogenic silica, is suggested now to be a general family trait of the Marattiaceae.
Key words - biogenic silica, calcium oxalate, Marattiaceae
RESUMO – (Caracterização de biominerais por espectroscopia infravermelha em Marattiaceae). Mostras de frondes dos fetos eusporangiados dos gêneros Angiopteris, Christensenia, Danaea e Marattia (Marattiaceae), foram investigadas por
espectroscopia infravermelha sob diferentes condições experimentais. Os resultados conirmaram relatos anteriores sobre
a acumulação de sílica biogênica (sio2), em tecidos desses fetos e também mostraram, pela primeira vez, a presença de oxalato de cálcio nesse grupo de plantas, provavelmente na forma de weddellite. a capacidade de mineralizar sio2, para produzir e acumular sílica biogênica, sugere que esta característica seja uma propriedade genérica dos membros da família Marattiaceae.
Palavras-chave - Marattiaceae, oxalato de cálcio, silica biogênica
1. universidad nacional de la Plata, Facultad de Ciencias Exactas, Centro de química inorgánica (Cequinor/Conicet,unlP), Casilla de Correo 962, 1900 la Plata, argentina.
2. Facultad de Ciencias naturales y Museo de la Plata, laboratorio de Estudios de anatomía Vegetal Evolutiva y sistemática (leaves), 64 entre 120 y diagonal 113, B1904 DZB la Plata, argentina. 3. Corresponding author: baran@quimica.unlp.edu.ar
Introduction
the presence and wide distribution of biogenic
silica (sio2, opal) in the plant kingdom is well known
(Blackman 1971, Bertoldi de Pomar 1975, arnott 1982, Perry 1989, 2003, Monje & Baran 2004).
silica bodies are found in cells of leaves and other parts of the plant body in several groups of vascular plants, such as Equisetaceae and Hymenophyllaceae, among the Pteridophyta, or as in genera and species of many angiospermous families, such as aristolochiaceae, loranthaceae, Podostemaceae, Cannaceae, Commelinaceae, and Poaceae (Haberlandt 1965), the latter being the most studied group due to the diversity of morphological cell types involved in the accumulation of silica.
Ferns are a group with scarce studies related to silica bodies. Recently, one of us has performed systematic
analyses of the sio2 accumulation in eusporangiate
ferns belonging to the Marattiaceae Bercht. & J. s. Presl
(Rolleri 1993, 2002, 2003, Rolleri et al. 1987, 1991,
2003, 2004). these studies were carried out through microchemical analysis using the phenol test (Johansen 1940), and also using polarized light microscopy to calculate mineralogical constants. they demonstrated that three of the ive marattiaceous genera, Angiopteris
Hoffm., Archangiopteris H. Christ & Giensenh. and
Marattia sw. always have opal idioblasts in the epidermis
of lamina of pinnae, pinnulae or segments, and also in cells of the inner parenchymatose tissues. the other two
genera, Christensenia Maxon and Danaea sm., lack of
specialized epidermal cells with opal inclusions (Rolleri
et al. 2003).
Most species belonging to the ive genera were also investigated by means of scanning electron
microscopy and EDaX microanalysis (Rolleri et al.
axes and laminae. Besides, although opal is absent in
the epidermal cells of Christensenia and Danaea, all
species of both genera bear small amounts of amorphous silica inclusions within the parenchymatose tissues of axes and fronds.
the previous studies point to the fact that, no matter where is the silica inally accumulated, all the marattiaceous genera may produce it, either abundant
and easily detected, as reported for Angiopteris,
Archangiopteris and Christensenia, or as small isolated
silica bodies, more or less dispersed in the internal tissues of the plant body. thus, the aim of the present work was to advance into a wider physicochemical characterization of these bioinorganic deposits by performing an infrared spectroscopic study of several species of the genera of the marattiaceous ferns, in order to attain more accurate information on the silica bodies present in genera with and without epidermal idioblasts.
Material and methods
Plant material – the following species were selected for analysis: Angiopteris evecta (Forst.) Hoffm., A. pruinosa
Kunze, Christensenia aesculifolia (Bl.) Maxon subsp.
aesculifolia, C. aesculifolia (Bl.) Maxon subsp. P. korthalsii
(de Vriese) Rolleri, Danaea nodosa (l.) sm., D. moritziana
C. Presl, Marattia laevis sm., M. sylvatica (Bl.), and M. weinmanifolia liebm. they came from herborized specimens obtained from not poisoned material of the u.s. national Herbarium. samples were taken from plants collected in different locations of the geographical area of each species. a detailed citation list of specimens under study can be found in Rolleri et al. (2004).
treatment of plant material – the plant samples were treated as follows before their spectra were recorded: a) dry material was pulverized in a vibrating mill during different periods of time, up to 15 hours, using agate sample holders and balls; b)
the manually dispersed, dry material, was heated in a mufle
furnace in air, at different temperatures, up to 400 °C, as described by lanning et al. (1958); c) the dry material was treated with concentrated nitric acid and carefully heated over a water bath in air, as described by Kamatani (1971).
infrared spectra – the spectra were recorded on a FtiR-Bruker iF66 spectrophotometer in the range between 4000 and 400 cm-1, using the KBr pellet technique (ca. 4 mg of the powdered sample dispersed in 100 mg of KBr) (Drago 1965, Baran 2005). the experiments performed with each sample of all the investigated fern taxa showed a similar behavior. since all iR spectra resulted comparable, and only minor differences in relative band intensities were observed, only those obtained with the A. pruinosa sample were illustrated in detail.
terminology – the chemical and spectroscopic terminology was taken from Baran (1995, 2005) and Monje & Baran (2004), terms on ferns come from Rolleri et al. (1991, 2004) and lellinger (2002).
Results
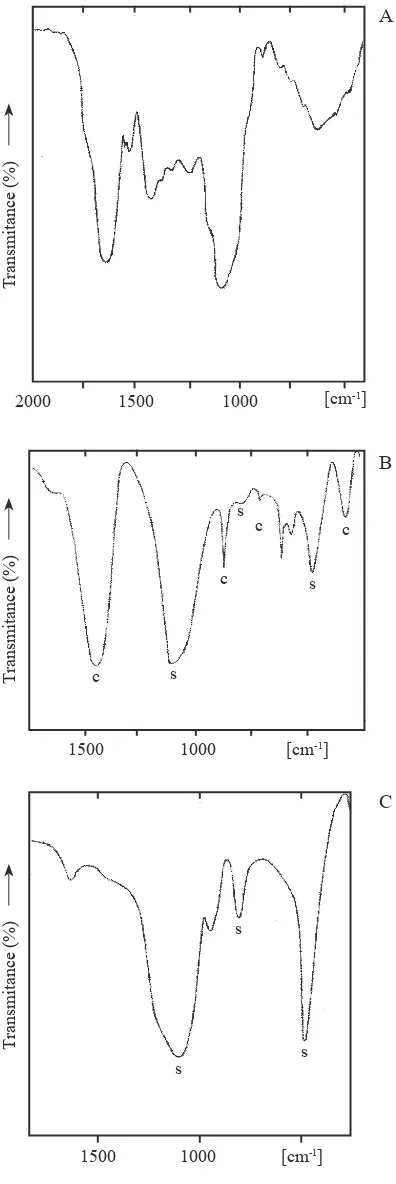
the iR spectrum of a sample milled during 15 hours, is shown in igure 1A for the most interesting spectral
range between 2000 and 450 cm-1. the intense band
located at 1641 cm-1 is characteristic of calcium oxalate
(corresponding to the antisymmetric oxalate stretching mode (Monje & Baran 1996, 1997)). the corresponding symmetric stretching is probably the weak band found
at 1340 cm-1 partially overlapped by the unidentiied
medium intensity band centered at 1427 cm-1. the
other strong band, centered at 1072 cm-1 is one of the
characteristic biogenic silica bands (Kamatani 1971, Perry 1989). the remaining broad feature located
between 850 and 450 cm-1 includes both unresolved
oxalate and silica bands. in the higher frequency side of the spectrum (not shown) a very broad and undeined
band, centered at about 3380 cm-1 is observed. this band
is related to o-H stretching modes of water and of si-oH groups.
Crystalline calcium oxalates have been found in two different hydration states in plants, either as
the monohydrate (whewellite, CaC2o4.H2o) or as
the dihydrate (weddellite, CaC2o4.2H2o) (arnott
1982, Monje & Baran 2004; Baran & Monje 2008). unfortunately, it was not possible to arrive to an unambiguous identiication of the chemical nature of the calcium oxalate found in these plants on the basis of the only relatively well deined oxalate IR
band (located at 1641 cm-1 in the A. pruinosa sample
and between 1641 and 1658 cm-1 in the spectra of
the other species) one could tentatively suggest the presence of weddellite (pure weddelite band is found at
1645 cm-1 whereas in whewellite it is found at 1622 cm
-1 (Monje & Baran, 1996, 1997)). Furthermore, the
low frequency spectral range which usually allows a clear differentiation between the two calcium oxalate forms (Monje & Baran 2002), appears partially
overlapped by one of the typical sio2 bands and is
highly undeined.
a clearly different iR spectrum was obtained when one of the milled samples, or a dry sample of the plant material, was slowly heated in a mufle furnace up to 400 °C and then maintained for 2 hours at this temperature (igure 1B). This spectrum shows the presence of calcite
of plane deformation, v2), 714 (in plane deformation, v4)
and 326 (lattice mode) cm-1 (Ross 1972)) and biogenic
silica (bands at 1100, 796 and 476 cm-1 (Kamatani 1971,
Perry 1989)).
this thermally generated product was treated with diluted HCl, then washed several times with distilled water and dried at 100 °C. the recorded iR spectrum of this new sample, shown in igure 1C, is different from that found in igure 1B and only the three characteristic absorptions of biogenic silica, together with a weak unidentiied feature at 941 cm-1, remain in the spectrum.
this spectrum is identical to those measured in other biogenic silica samples (Kamatani 1971).
it is noteworthy the fact that after heating the milled sample at 200 °C during short periods of time the spectra show oxalate, carbonate, and silica bands. Besides, the treatment of the plant material with nitric acid generates a solid which iR spectrum presents only the characteristic opal bands. it become evident that organic matter, together with the oxalate deposits, are both destroyed during the treatment with a strong oxidizing acid, and
only sio2, a species which is stable in acidic media, is
fully recovered.
Discussion
The performed spectroscopic study conirms, in an independent way, the previously established existence of opal deposits in marattiaceous ferns. these measurements also show, for the irst time, the presence of calcium oxalate in ferns. the presence of this biomineral is additionally supported by the chemical behavior of the investigated material. the heating of a calcium oxalate/ opal mixture generates calcite, whereas opal remains unchanged, as was spectroscopically observed, and may be due to the following reaction:
CaC2o4.nH2o + sio2.mH2o → CaCo3 + Co + (n + m)H2o + sio2
the subsequent treatment of the obtained solid residue with HCl, decomposes the generated carbonate:
sio2 + CaCo3 + 2 HCl → sio2 + CaCl2 + Co2 + H2o
and, after the washing, only sio2 is recovered, as was
also spectroscopically conirmed.
the heating of the plant material at lower temperatures and for short periods of time generates a mixture which contains part of the original biominerals
together with CaCo3, which is produced by the partial
decomposition of the oxalate.
Figure 1. a. FtiR spectrum of a powdered sample of
Angiopteris pruinosa dry fronds. B. FtiR spectrum of dry fronds of Angiopteris pruinosa heated during 2 h at 400 °C. C. FtiR spectrum of a heated sample, after treatment with
HCl. (band identiication: C = calcite bands; S = SiO2 (opal) bands).
a
B
C
t
ransmitance (%)
t
ransmitance (%)
t
ransmitance (%)
c s
c s
c
s c
s s
s
2000 1500 1000
1500 1000
1500 1000
[cm-1]
[cm-1]
[image:3.595.73.271.69.661.2]although it was not possible to identify unambiguously the chemical nature of the calcium oxalate accumulated by these ferns, weak evidence derived from the position of the antisymmetric oxalate stretching vibration points out to the presence of
weddellite, (CaC2o4.2H2o).
the ability to biomineralize sio2 and to produce opal
was considered a diagnostic trait only at the generic level
in the Marattiaceae (Rolleri et al. 2003, 2004), but after the
results of the present study we suggest to consider it a family character, shared by all ive genera. While the distribution
of epidermal idioblasts in Angiopteris, Archangiopteris
and Marattia varies from solitaire, groups from 2 to 6
idioblasts, or extensive groups of these cells containing
opal (idioblast areas, Rolleri et al. 2003), the cells of soft
tissues near costae and secondary veins of pinnae of species belonging to all genera bear isolated, generally small, silica bodies. these bodies were previously observed through longitudinal and transversal sections of axes and pinnae, and also after clearing the foliar tissues of laminae. The now performed IR spectroscopic study conirms
that these bodies are also sio2 inclusions. the internal
amorphous crystals are smaller than those accumulated in the epidermis, and more dificult to be observed than the epidermal silica, mostly composed of inclusions as large as the idioblasts themselves. since all genera also have opal in scattered cells of inner parenchymatous tissues, specially in those situated close to the vascular bundles, the results obtained by IR spectroscopy conirm one of the outstanding characters of the family.
acknowledgements – the authors are indebted to Dr. David lellinger, formerly Curator of Ferns of the u.s. national Herbarium, who gave the authorization to carry out this kind of studies with samples of herbarium material. this work has been supported by the Consejo nacional de investigaciones
Cientíicas y Técnicas de la República Argentina (Conicet)
and the universidad nacional de la Plata. the authors are members of the Research Career of Conicet.
References
aRnott, H.J. 1982. three systems of biomineralization in plants with comments on the associated organic matrix.
In Biological mineralization and demineralization (G.H. nancollas, ed.). springer, Berlin, p.199-218.
BaRan, E.J. 1995. química bioinorgánica, Mc-Graw Hill interamericana de España s.a., Madrid.
BaRan, E.J. 2005. applications of vibrational spectroscopy to the investigation of plant material. In advances in
plant physiology (H. Hemantaranjan, ed.). Scientiic
Publishers, Jodhpur, v.8, p.365-392.
BaRan, E.J. & MonJE, P.V. 2008. oxalate biominerals.
In Metal ions in life sciences (a. sigel, H. sigel &
R.K.O. Sigel, eds.). J. Wiley & Sons, Chichester, v.4,
p.219-254.
BERtolDi DE PoMaR, H. 1975. Los silicoitolitos: sinopsis de su conocimiento. Darwiniana 19:173-206. BlaCKMan, E. 1971. opaline silica bodies in the range
grasses of southern alberta. Canadian Journal of Botany 49:769-781.
DRaGo, R.s. 1965. Physical methods in inorganic chemistry, Reinhold Publishing Corporation, new York.
HaBERlanDt, G. 1965. Physiological plant anatomy, today & tomorrows Book agency, new Delhi. JoHansEn, D.a. 1940. Plant microtechnique. McGraw-Hill,
new York.
KaMatani, a. 1971. Physical and chemical characteristics of biogenous silica. Marine Biology 8:89-95.
LANNING, F.C., PONNAYA, B.W.X. & CRUMPTON,
C.F. 1958. the chemical nature of silica in plants. Plant Physiology 33:339-343.
lEllinGER, D.B. 2002. a modern multilingual glossary of taxonomic Pteridology. Pteridologia no 3, american
Fern Society, Washington.
MonJE, P.V. & BaRan, E.J. 1996. on the formation of weddellite in Chamaecereussilvestrii, a Cactaceae from northern argentina. Zeitschrift für naturforschung 51c: 426-428.
MonJE, P.V. & BaRan, E.J. 1997. on the formation of whewellite in the Cactaceae species Opuntia microdasys. Zeitschrift für naturforschung 52c:267-269.
MonJE, P.V. & BaRan, E.J. 2002. Characterization of calcium oxalates generated as biominerals in cacti. Plant Physiology 128:707-713.
MonJE, P.V. & BaRan, E.J. 2004. Plant biomineralization.
In advances in plant physiology (H. Hemantaranjan,
ed.). Scientiic Publishers, Jodhpur, v.7. p.395-410.
PERRY, C.C. 1989. Chemical studies of biogenic silica.
In Biomineralization – chemical and biochemical
perspectives (S. Mann, J. Webb & R.J.P. Williams, eds.). Verlag Chemie, Weinheim, p.223-256.
PERRY, C.C. 2003. Siliciication: the process by which organisms capture and mineralize silica. In Reviews in mineralogy & geochemistry 54. Biomineralization (P.M.
Dove, J.J. De Yoreo & S. Weiner, eds.). Mineralogical Society of America, Washington D.C., p.291-327.
RollERi, C.H. 1993. Revision of genus Christensenia. american Fern Journal 83:3-19.
RollERi, C.H. 2002. Caracteres diagnósticos y taxonomía del género Angiopteris Hoffm.(Marattiaceae) i. los caracteres. Revista Museo la Plata, Botánica 15: 23-49.
RollERi, C.H., DEFERRaRi, a.M. & CiCCiaRElli, M. 1987. Epidermis y estomatogénesis en Marattiaceae (Marattiales). Revista Museo la Plata, Botánica 14: 124-147.
RollERi, C.H., DEFERRaRi, a.M. & laVallE, M.C. 1991. Epidermal morphology of the pinnae of
Angiopteris, Danaea and Marattia. american Fern Journal 81:44-62.
RollERi, C.H., laVallE, M.C., MEnGasCini, a. & RoDRiGuEZ, M. 2003. sistemática de helechos eusporangiados (Marrattiaceae-Marattiales). Revista Museo la Plata, Botánica 16:49-64.
RollERi, C.H., laVallE, M.C., MEnGasCini, a. & RoDRiGuEZ, M. 2004. idioblastos de ópalo en helechos maratiáceos. Physis (Buenos aires) C 59:13-21. Ross, s.D. 1972. inorganic infrared and Raman spectra,